
The units of half-life are always time (seconds, minutes, years, etc.). In other words, it is the lifetime of half the radioactive isotopes in a system. modified from Webster's Third International Dictionary, Unabridged The time required for one-half of the radioactive (parent) isotopes in a sample to decay to radiogenic (daughter) isotopes. In order to determine the age of a geologic material, we must understand the concept of half-life. Radioactive elements can be used to understand numerical age of geological materials on time scales as long as (and even longer than) the age of the Earth. Problem solving in the geosciences was forever changed with the discovery of radioactivity. Remind them that geologists only use certain radioactive isotopes to date rocks. Samarium (Sm) has 7 naturally occurring isotopes (3 are radioactive). However, oxygen atoms can have between 8 and 10 neutrons in the nucleus and therefore the isotopes of oxygen have atomic masses of 16, 17, and 18 a.m.u.(and none are radioactive!). For example, all atoms of oxygen have 8 protons in the nucleus and hence have an atomic number of 8. Isotopes of an element are atoms that all have the same atomic number (or number of protons in the nucleus) but have different atomic masses (hence different numbers of neutrons in the nucleus). They may ask, "What's the difference between an isotope and an atom?" Another way of explaining it is that when geologists talk about isotopes, they are talking about one element of differing masses. That definition may not mean anything to them. "A variety of a chemical element (strictly, of one particular element) which is distinguished from the other varieties of the element by a different mass number but shares the same atomic number and chemical properties (and so occupies the same position in the periodic table)." The Oxford English Dictionary defines an isotope as: Often students struggle with the concept of isotopes. If they can begin to comprehend that it is random and spontaneous, they end up feeling less nervous about the whole thing. Often students get bogged down in the fact that they don't "understand" how and why radioactive elements decay and miss the whole point of this exercise. Statistical probablity is the only thing we can know exactly. Students often struggle with this concept therefore, it should be stressed that it is impossible to know exactly when each of the radioactive elements in a rock will decay. Radioactivity and radioactive decay are spontaneous processes. Radioactivity: A steady but unpredictable (spontaneous) process 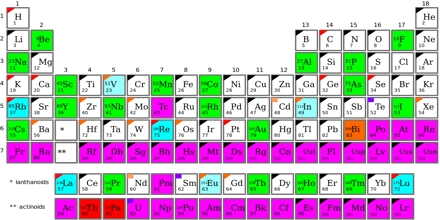
the spontaneity (or randomness) of radioactive decay,.Generally, there are four main concepts that students struggle with when thinking about radioactive decay: Jump down to: Isotopes | Half-life | Isotope systems | Carbon-14 | Examples & Exercises Essential Concepts Wenner, Geology Department, University of Wisconsin-Oshkosh How Does Radioactive Decay Work? created by Jennifer M. Quantitative concepts: exponential growth and decay, probablility
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
